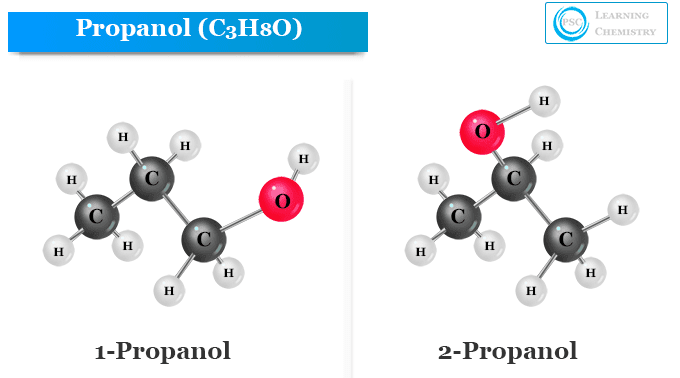
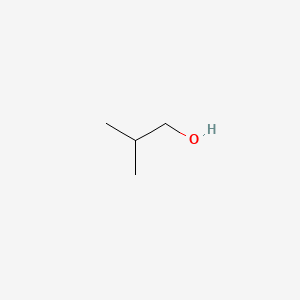
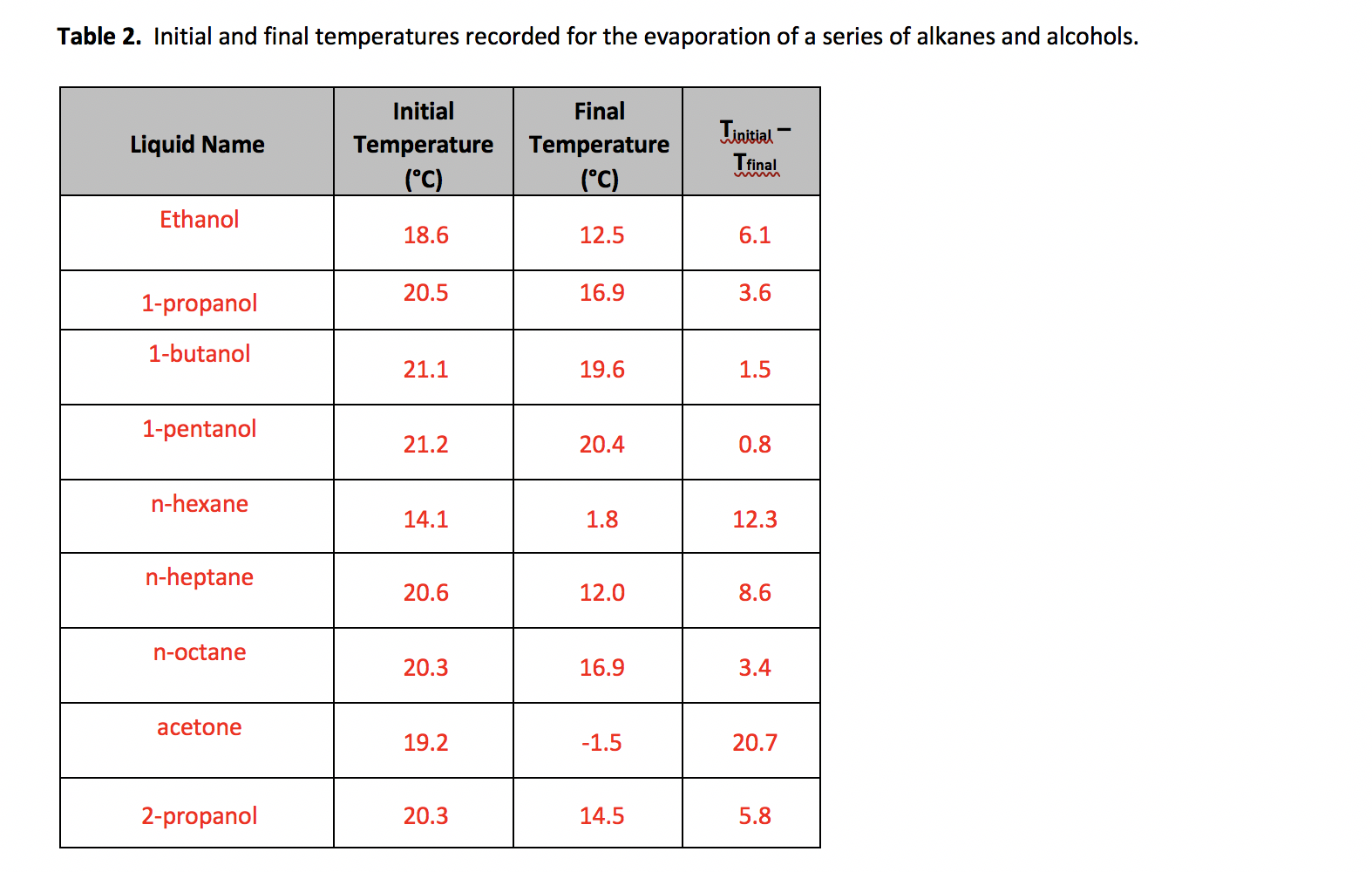
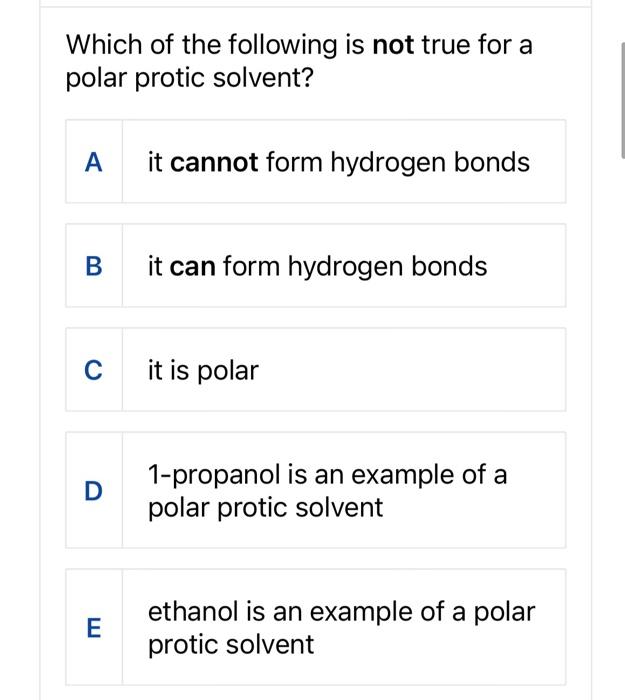
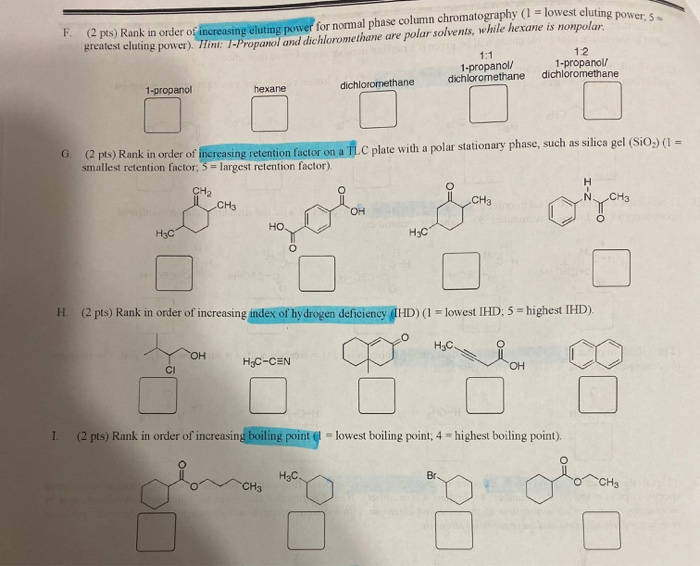
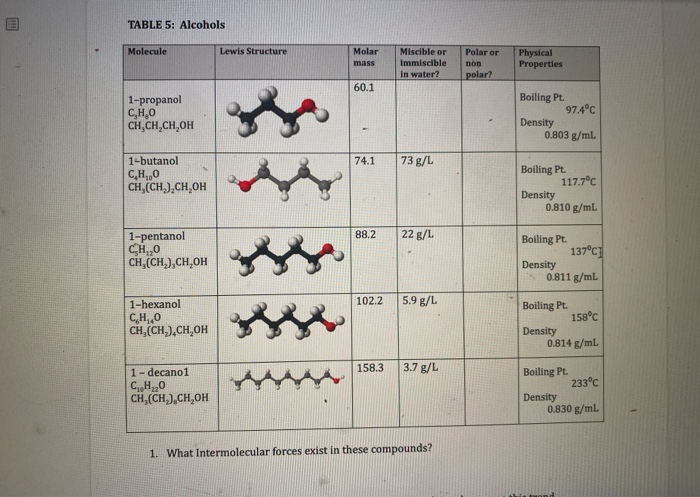
Is 1-propanol an ionic, molecular nonpolar, or molecular polar compound? What intermolecular forces are present? | Homework.Study.com

SOLVED: An abbreviated Lewis Structure of propanol is shown below (lone pairs are omitted) In which of the following solvents would propanol be most soluble? H H H 4-4 4-0-H water (H,O)

Dependence of characteristic concentration on Snyder's polarity index... | Download Scientific Diagram

Pharmacognosy for all - Polarity index table of commonly used solvents To calculate the polarity index of a mixture of solvents : Polarity index of the mix.= (Polarity index of solvent 1 *

SOLVED: Table Relative polarity of common mobile phases: Acetic acid Water Methanol Ethanol Pyridine Propanol Acetone [ Ethyl acetate Diethyl ether 1 Ten- Butyl mcthyl cther Dichloromethane Chloroform" a lene Carbon tetrachloride