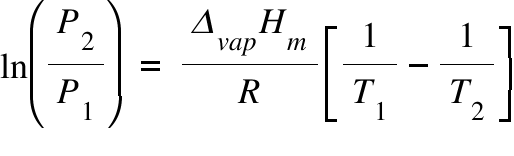One moles of strem is compressed reversibly of water at boiling point 100^(@)C. The heat of vapourisation of water at 100^(@)C and 1atm is 540cal g^(-1). Calculate DeltaU and DeltaH.

SOLVED:Calculate the heat change at 100^∘ C for each of the following and indicate whether heat was absorbed or released: a. calories to vaporize 10.0 g of water b. joules to vaporize